Filter
37
Featured
4
5
Language
Document type
9
5
5
5
3
3
3
2
1
1
Countries
3
3
3
3
1
1
Authors & Publishers
Publication Years
Category
22
2
1
1
Important Update - Changes to WHOs Prequalification of IVD Assessment Procedure (from 2026)
Starting in 2026, WHO will introduce a new prequalification assessment procedure. The performance evaluation will no longer be part of the prequalification assessment and will become a separate procedure.
R
...
The course is divided into four thematic modules:
1. General information; 2. Laboratory diagnosis; 3. Plasmodium species identification and parasite counting; 4. Microscopy quality assurance. To update and review the basic concepts of epidemiology and laboratory diagnosis of malaria, as well as the
...
El curso de autoaprendizaje Diagnóstico microscópico de la malaria está dirigido a los microscopistas y al personal de laboratorio de la Región de las Américas, con el fin de refrescar los conocimientos básicos relativos a la malaria y su diagnóstico a través del uso de la microscopia, inclu
...
The new tests address this issue by targeting a different parasite protein (pf-LDH) that the malaria parasite cannot easily shed. They provide a reliable, quality-assured alternative where HRP2-based tests are failing. WHO now recommends that countries switch to these alternative RDTs when more than
...
Technical specifications series for submission to WHO prequalification: diagnostic assessment;TSS-3
Lecture. Accessed July, 2025
Version 46 22 Dec 2025. his document provides an overview of malaria rapid diagnostic tests (RDTs) for Principal Recipients (PRs) of Global Fund grants, indicating their eligibility for procurement under the Global Fund's Quality Assurance Policy. The included products have been assessed and approve
...
Relapsing malaria caused by Plasmodium vivax parasites poses a significant challenge to global malaria elimination efforts. About one third of the population remains at risk of contracting P. vivax malaria, and 85% of P. vivax infections stem from reactivated latent parasites, leading to chronic ana
...
The overall objective of this approach is to accelerate efforts toward malaria elimination by increasing
access to early diagnosis and treatment. The specific objectives are to: Minimize the barriers that the population faces in accessing timely, quality diagnosis and treatment. Reduce the time bet
...
for health care providers
This document is a guide defining requirements for quality and safety for malaria rapid diagnostic testing services to safeguard the quality of the results, the safety of the operators and patients and that of the environment for use by national malaria control programmes, regulators, implementers a
...
The 2022 report reviews the global malaria diagnostics market and technological landscape to support Unitaid’s 2023–2027 strategy for quality malaria case management. The report highlights the stalled progress of malaria control efforts, the gaps in access to diagnostics and the public health im
...
second edition
procédures : guide méthodologique
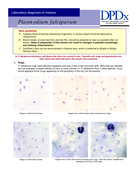
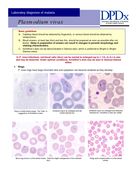
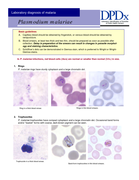
Estas Pranchas para o diagnóstico microscópico da malária servem como guia para laboratoristas e técnicos de campo responsáveis por diagnóstico microscópico da malária pelo método de Giemsa. Também devem ser úteis para professores e alunos de disciplinas correlatas. As pranchas mostram fo
...
Segunda Edição. Este manual (Parte I dos módulos de capacitação em Bases do diagnóstico microscópico da malária) ajudará os participantes durante seu treinamento em diagnóstico microscópico da malária humana. O manual foi concebido como base para um treinamento formal de quatro a cinco s
...
O volume de capacitação em Bases do diagnóstico microscópico da malária é composto por dois módulos, que servem como uma estrutura para que os instrutores montem o curso. Ele fornece as informações mínimas necessárias para capacitar alunos em diagnóstico microscópico da malária pelo m�
...