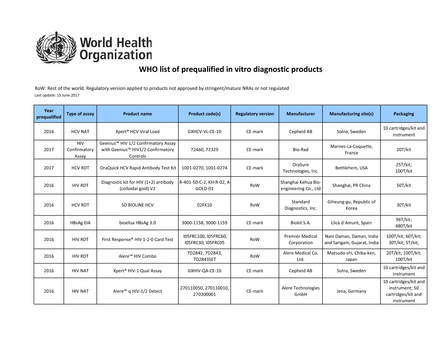
WHO list of prequalified in vitro diagnostic products
recommended
Important Update - Changes to WHOs Prequalification of IVD Assessment Procedure (from 2026)
Starting in 2026, WHO will introduce a new prequalification assessment procedure. The performance evaluation will no longer be part of the prequalification assessment and will become a separate procedure.
Rapid advances in development of medical devices are generating challenges in quality assurance for manufacturers and regulators, and in both quality assurance and product selection for procurers. Launched in 2010, WHO prequalification of in vitro diagnostics provides a valuable service to each of these groups.