Filter
26
Featured
5
Language
Document type
10
5
4
2
1
1
1
1
1
Countries
3
3
2
2
2
1
1
1
1
1
1
1
Authors & Publishers
Publication Years
Category
10
1
COVID-19 vaccine tracker and landscape
recommended
The COVID-19 vaccine tracker and landscape compiles detailed information of each COVID-19 vaccine candidate in development by closely monitoring their progress through the pipeline.
The COVID-19 vaccine tracker:
Provides summary tables of COVID-19 vaccine candidates in both clinical and pre-
...
Interim Guidance October 2022. This addendum addresses some of the methodological aspects of VE evaluations that have been learned during the past year, as well as those that have become relevant in the current epidemiological setting of the COVID-19 pandemic. For some of the COVID-19 vaccine method
...
This chronology of facts has challenged public health systems worldwide and regulatory bodies are no exception. Regulatory authorities with mechanisms in place to authorize the use of investigational products had to development guidelines and procedures, create task forces and alliances to maximize
...
29 April 2022
What are the new vaccines in the pipeline? What are the challenges to turning vaccines into vaccination ? What timelines can we expect for future vaccines? WHO’s Dr Katherine O’Brien explains in Science in 5.
The European Medicines Agency (EMA) is evaluating potential COVID-19 treatments and vaccines to enable promising medicines to reach patients as soon as possible. It is also interacting with medicine developers and making use of real-world data to monitor the safety and effectiveness of medicines use
...
Ausführliche Informationen zu klinischen Prüfungen und Risiken für bestimmte Gruppe sowie zu Wechselwirkungen und Nebenwirkungen.
Ausführliche Informationen zu klinischen Prüfungen und Risiken für bestimmte Gruppen sowie zu Wechselwirkungen und Nebenwirkungen von Impfstoffen und Arzneimitteln zu COVID-19 und SARS-CoV-2
COVID-19 Vaccine Resource Center
recommended
A collection of resources on Covid-19 vaccines, including frequently asked questions, continuing medical education, published research, and commentary.
Vaccines
recommended
COVID Reference 23 May 2021
February 2021.
Improving our response to the ongoing COVID-19 pandemic in Africa requires regularly updated information, constant innovation, and considerable support towards research and development (R&D) for priorities that respond to the African realities. Shaping the research agenda and stimula
...
21 Dec 2021. Slideset updated regularly to include the latest data and guidance on COVID-19 vaccines, including implications of emerging SARS-CoV-2 variants
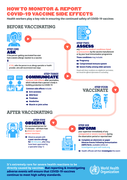
Evaluation of COVID-19 vaccine effectiveness
17 March 2021
Many critical questions remain about the effectiveness of COVID-19 vaccines in real-world settings. These questions can only be answered in post-introduction vaccine effectiveness studies.This guidance document outlines an approach to leverage existing surveillance systems for Severe Acute Respirato
...
Vacunas
recommended
COVID Reference 23 May 2021
This tracker, developed by the Vaccine Centre at the London School of Hygiene & Tropical Medicine, will follow COVID-19 vaccine candidates as they progress through the development pipeline. We will update it weekly. An overview of the different vaccine types as well as the phases of clinical develo
...