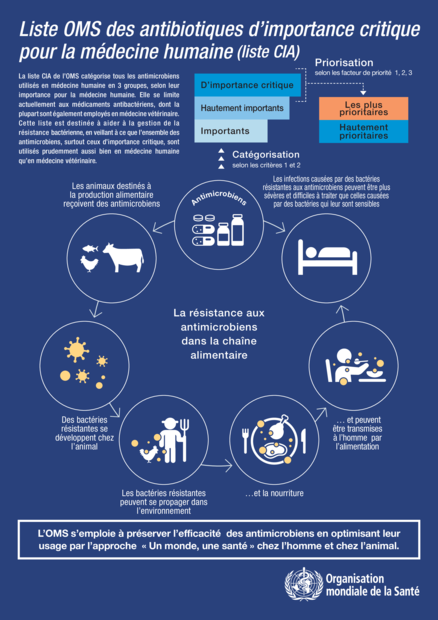
Liste OMS des antibiotiques d’importance critique pour la médecine humaine
La liste CIA de l’OMS catégorise tous les antimicrobiens utilisés en médecine humaine en 3 groupes, selon leur importance pour la médecine humaine. Elle se limite actuellement aux médicaments antibactériens, dont la plupart sont également employés en médecine vétérinaire.Cette liste est destinée à aider à la gestion de la résistance bactérienne, en veillant à ce que l’ensemble des antimicrobiens, surtout ceux d’importance critique, sont utilisés prudemment aussi bien en médecine humaine qu’en médecine vétérinaire.