Filter
417
Text search:
vials
Featured
69
112
Language
Document type
180
105
39
38
18
13
10
9
3
1
1
Countries / Regions
50
12
12
11
9
9
8
8
8
8
6
6
5
5
5
5
4
4
4
3
3
3
3
2
2
2
2
2
2
2
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
Authors & Publishers
Publication Years
Category
154
72
29
22
17
15
9
Toolboxes
59
44
25
25
20
16
14
13
11
10
7
6
5
5
5
5
4
3
3
1
1
1
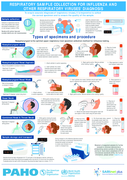
Respiratory sample collection for Influenza and other respiratory viruses diagnosis - Infographic
Presentation OPT-SMC malaria vaccine workshop - Jan 2023
The Practical Approach to Emergencies in the Pregnant Mother, Newborn infant and Child. Provder Manual
This guide provides a comprehensive overview of essential information related to immunization, including technical information about vaccines, a review of immunization program management best practices, guidance on the delivery of immunization services, monitoring and evaluation, disease surveillanc
...
First Global Patient Safety Challenge Clean Care is Safer Care
The WHO Guidelines on Hand Hygiene in Health Care provide health-care workers (HCWs), hospital administrators and health authorities with a thorough review of evidence on hand hygiene in health care and specific recommendations to impro
...
Training module on malaria control
These guidelines were developed through an interagency process with participation by United Nations (UN) organiza-
tions, nongovernmental organizations, the International Red Cross and Red Crescent Movement and the International
Organization for Migration.
Training Manual
Maintaining proper storage conditions for health commodities is vital to ensuring their quality. Product expiration dates are based on ideal storage conditions and protecting product quality until their expiration date is important for serving customers and conserving resources. Guidelines for the S
...
MSF Essential Drugs - Practical Guidelines 2022
recommended
Practical guide intended for physicians, pharmacists, nurses and medical auxiliaries. This guide is not a dictionary of pharmacological agents. It is a practical manual intended for health professionals, physicians, pharmacists, nurses and health auxiliaries invoved in curative care and drug managem
...
Surgical Care At The District Hospital
recommended
Promotion of the quality of clinical care through the identification, promotion and standardization of appropriate procedures, equipment and materials, particularly at district hospital level.
2nd edition
Snakebites are well-known medical emergencies in many parts of the world, especially in rural areas. Agricultural workers and children are the most affected. The incidence of snakebite mortality is particularly high in South-East Asia. Rational use of snake anti-venom can substantially reduce mortal
...
Best Practices in health care waste management
recommended
Examples from four Philippine Hospitals
These guidelines are designed for ICRC and other health professionals – nurses, midwifes, doctors – who either lack experience in antenatal care or are not used to working in countries where medical infrastructure is underdeveloped or non-existent
WHO practical guidelines. 2nd edition