Filter
629
Text search:
Polymerase
Chain
Reaction
(PCR)
Featured
106
191
Language
Document type
327
128
80
35
21
17
7
6
5
1
1
1
Countries / Regions
29
22
19
17
17
17
14
13
11
11
11
10
10
9
9
8
8
7
7
7
6
6
6
5
5
5
4
4
4
4
3
3
3
3
3
3
2
2
2
2
2
2
2
2
2
2
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
Authors & Publishers
Publication Years
Category
239
101
29
26
14
10
8
Toolboxes
74
72
40
37
35
30
26
19
17
10
9
7
7
5
4
3
3
2
2
2
1
1
Interium guidance, 25 June 2021Timely and accurate diagnostic testing is an essential tool in preventing and controlling the spread of COVID-19. This document describes recommendations for national testing strategies and the use of PCR and rapid ant
...
PLoS ONE 13(8): e0202499. https://doi.org/10.1371/journal.pone.0202499
This was a school-based cross-sectional study conducted in 2015 among 305 school children aged 7–16 years from two primary schools located in Ilemela and Magu Districts, north-western Tanzania. Single stool and urine samples w
...
i. A person who is a contact of a probable or confirmed mpox case in the 21 days before the onset of signs or symptoms, and who presents with any of the following: acute onset of fever (>38.5°C), headache, myalgia (muscle pain/body aches), back pain, profound weakness or fatigue.
OR
ii. A per
...
The National Guidelines for HIV-1 Viral Load Laboratory Testing support plans to scale up viral load (VL) testing to reach the 90-90-90 targets in India. This phased scale-up includes the setup of 70 additional VL testing laboratories nationally. These guidelines include laboratory design considerat
...
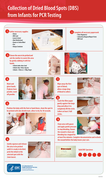
This clinical job aid provides health care workers with information on how to collect specimens for early infant diagnosis on dried blood spots, as well as drying and packaging for transport.
Au total, 18 laboratoires de 13 pays ont participé aux quatre cycles d'AQE : 10 laboratoires de huit pays africains endémiques, dont quatre ont participé aux quatre cycles et trois à trois cycles. Les résultats globaux ont montré que la performance médiane de ces laboratoires s'est amélioré
...
Diagnostik zum Nachweis von COVID-19
recommended
Im Kapitel zu Diagnostik zum Nachweis von COVID-19 bekommen Sie ausführlich die verschiedenen Testmethoden erklärt, es können Videos zu PCR, RAchenabstrich, etc. aufgerufen werden
PLoS One. 2012; 7(4): e29656.
Published online 2012 Apr 20. doi: 10.1371/journal.pone.0029656
Disease outbreak news, 21 November 2019
Towards Malaria Elimination
Acta Trop. 2020 Mar:203:105289. doi: 10.1016/j.actatropica.2019.105289
The role of malacological surveys to identify potential transmission sites for schistosomiasis control in this era of
mass drug administration have received little attention. In that context, the present study was conducted to
...
Trop Med Int Health. 2015 Apr; 20(4): 448–454. Open Access
Available in English, French, Spanish, Chinese, Russian and Arabic
https://apps.who.int/iris/handle/10665/337832
This document provides interim guidance on the prevention, identification and management of health worker infection in the context of COVID-19. It is intended for occupational health departments, infection prevention and control departments or focal points, health facility administrators and public
...