Filter
2222
Text search:
congo
Featured
218
684
Language
Document type
1141
488
195
125
106
55
46
26
17
9
8
3
3
Countries
486
152
113
111
104
101
101
93
87
84
75
74
60
55
53
48
45
42
40
39
39
36
35
28
28
22
22
22
20
19
18
16
15
15
15
14
13
13
12
12
11
11
11
10
10
10
9
9
9
8
8
7
7
6
6
6
5
5
5
5
5
4
4
4
4
4
4
4
3
3
3
3
3
3
3
3
3
3
3
3
2
2
2
2
2
2
2
2
2
2
2
2
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
Authors & Publishers
Publication Years
Category
715
166
163
128
114
36
15
1
Toolboxes
351
195
115
104
78
75
73
73
72
64
62
51
50
37
35
27
26
26
25
20
20
17
9
9
7
2
2
7 Febr. 2021
As COVID-19 continues to wreak havoc in countries – decimating people’s livelihoods, and leaving health systems struggling to provide healthcare and vaccines for the entire population - governments and donors should look to the Church as a partner. The essential Church networks, tr
...
Many African countries were amongst the most rapid to respond to the emerging threat of COVID-19, implementing large-scale interventions at very early stages of their epidemic. As demonstrated in this document using very simple models, this rapid mobilization and timeliness of implementing control m
...
In Central and West Africa, regions together comprising 27 countries and 605 million people, the average person is exposed to particulate pollution levels that are more than 4 times the World Health Organization’s (WHO) guideline of 5 μg/m³1. If these particulate pollution levels persist, averag
...
Cholera English Information
recommended
MEDBOX Issue Brief 26. Cholera is an acute diarrhoeal infection caused by eating or drinking food or water that is contaminated with the
bacterium Vibrio cholerae. Cholera remains a global threat to public health and is an indicator of inequity and lack of
social development. Researchers have esti
...
This is the seventeenth annual publication of the Global Hunger Index (GHI), a report jointly published by Concern Worldwide and Welthungerhilfe.
The 2022 Global Hunger Index (GHI) brings us face to face with a grim reality. The toxic cocktail of conflict, climate change, and the COVID-19 pandemi
...
Uganda hosts approximately 1.1 million refugees making it Africa’s largest refugee hosting country and one of the five largest refugee hosting countries in the world. Most recently, throughout 2016- 2018, Uganda was impacted by three parallel emergencies from South Sudan, the Democratic Republic o
...
DNDi is now striving to make fexinidazole available to the majority of people who have T.b. gambiense sleeping sickness. We are supporting a three-year access and pharmacovigilance study that began in 2020 and have so far carried out in-country training of relevant staff in 250 hospitals and
...
During Epidemiological week (Epiweek) 5, 20 countries in the WHO African region (WHO AFR) contributed virological data for analysis - Algeria, Burkina Faso, Cameroon, Central African Republic, Côte d’Ivoire, Democratic Republic of the Congo, Ethi
...
Mpox is a zoonotic disease caused by a double-stranded DNA virus that belongs to the Orthopoxvirus genus of the Poxviridae family. The disease presents with symptoms similar to smallpox but with a lesser severity. It was first discovered in 1958 when two outbreaks of a poxlike disease occurred in co
...
The Malaria Ministerial Conference, co-hosted by WHO and the Government of Cameroon on 6 March 2024, brought together more than 400 stakeholders, including Ministers of Health and senior representatives from the African countries hardest hit by malaria, global health leaders, scientists, civil socie
...
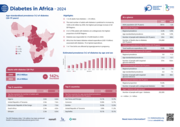
Diabetes poses an increasingly serious health challenge in Africa. Currently, 25 million adults aged 20–79 are living with diabetes, and this figure is projected to increase by 142% to 60 million by 2050 — the highest regional increase worldwide. However, 73% of cases remain undiagnosed, and hea
...
The 2024 World Malaria Report shows that the malaria burden remains overwhelmingly concentrated in Africa. The continent accounted for 94% of global cases and 95% of malaria-related deaths in 2023. Although the number of malaria cases increased globally from 204 million in 2000 to 246 million in 202
...
The West African Ebola virus disease (EVD) outbreak in 2014 emerged in a region in Africa that had previously not reported any EVD outbreak. Subsequently, the outbreak rapidly spread and was protracted in the three Mano River basin countries (Guinea, Sierra Leone and Liberia), with spillover cases i
...
This is the seventeenth annual publication of the Global Hunger Index (GHI), a report jointly published by Concern Worldwide and Welthungerhilfe.
The 2022 Global Hunger Index (GHI) brings us face to face with a grim reality. The toxic cocktail of conflict, climate change, and the COVID-19 pandemi
...
After 100 years of chemotherapy with impractical and toxic drugs, an oral cure for human African trypanosomiasis (HAT) is available: Fexinidazole. In this case, we review the history of drug discovery for HAT with special emphasis on the discovery, pre-clinical development, and operational challenge
...
The Transformation Agenda (TA) ushered in an ambitious reform process intended to transform the World Health Organization (WHO) into an organization that is proactive, results-driven, accountable and which meets stakeholder expectations, towards transforming and improving public health services in t
...
Mpox continues to affect people around the world. A new framework released today by WHO will guide health authorities, communities and other stakeholders in preventing and controlling mpox outbreaks, eliminating human-to-human transmission of the disease, and reducing spillover of the virus from ani
...
L’orthopoxvirus simien ou virus de la variole du singe (MPXV), est un virus à ADN double brin qui appartient au genre Orthopoxvirus de la famille
des Poxviridés. Les poxvirus causent des maladies chez l’espèce humaine et beaucoup d’autres animaux ; l’infection se manifeste généralement
...
Integrated Outbreak Analytics (IOA) applies a multidisciplinary approach to understanding outbreak dynamics and to inform outbreak response. It aims to drive comprehensive, accountable, and effective public health and clinical strategies by enabling communities, and national and subnational health a
...
Epidemiol Infect. May 2007; 135(4): 610–621. . Please click on the website link and download the pdf-file. Open Access