Filter
4898
Text search:
medicines
Featured
519
1334
Language
4636
150
132
98
70
48
40
32
18
17
9
9
8
7
6
6
6
5
5
5
5
4
4
4
3
3
3
3
3
3
2
2
2
2
2
2
2
2
2
2
2
2
2
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
Document type
2350
853
738
298
236
149
109
67
33
30
16
11
6
1
Countries / Regions
244
229
150
116
115
104
95
94
94
89
87
81
76
75
74
74
74
72
72
70
53
51
47
42
41
40
38
36
33
30
29
27
25
25
24
24
24
23
23
23
23
23
22
21
20
18
18
16
16
14
12
11
11
11
10
10
10
10
10
9
9
9
9
9
7
7
7
7
6
6
6
6
6
5
5
5
5
5
5
5
4
4
4
4
4
4
4
4
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
Authors & Publishers
766
240
174
134
132
82
80
77
64
57
34
34
32
31
29
27
27
25
25
25
25
21
20
19
19
19
19
18
17
16
16
16
16
15
15
15
15
14
14
13
13
13
13
13
13
13
13
12
12
12
11
11
11
11
11
10
10
10
10
10
10
10
10
10
10
9
9
9
9
9
8
8
8
8
8
8
8
8
8
8
8
8
8
7
7
7
7
7
7
7
7
7
7
7
7
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
6
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
Publication Years
1584
2908
377
27
2
Category
2008
414
292
207
177
97
59
2
Toolboxes
370
296
291
288
273
251
159
145
133
128
111
110
106
94
91
77
64
51
43
39
20
19
12
11
7
1
GDF is the largest global provider of quality-assured tuberculosis (TB)
medicines, diagnostics, and laboratory supplies to the public sector.
Since 2001, GDF has facilitated access to high-quality TB care in over 130
countries, providing treatmen
...
ts to over 30 million people with TB and procuring
and delivering more than $200 million worth of diagnostic equipment
more
Researchers are devising a clinical-trial protocol to test three medicines in Africa's latest outbreak
Nature doi: 10.1038/d41586-018-06132-7
When an infection strikes, and medicines like antibiotics (and other antimicrobials) do not work, you can lose your entire stock of animals to disease. This also puts the health of you and your family at risk when the infection can spread between an
...
imals and people. Save lives and livelihoods by following the advice below starting today!
more
The Pharmacovigilance team in WHO aims to assure the safety of medicines and vaccines by ensuring reliable and timely exchange of information on safety issues, promoting pharmacovigilance activities throughout the Organization and encouraging partic
...
ipation in the WHO Programme for International Drug Monitoring. This text was developed in consultation with the WHO Collaborating Centre for International Drug Monitoring and the national pharmacovigilance centres participating in the WHO Programme for International Drug Monitoring.
more
Global UNIDO Project: Strengthening the local production of essential medicines in least developed and developing countries
In 2015, the United Nations set important targets to reduce premature
cardiovascular disease (CVD) deaths by 33% by 2030. Africa disproportionately
bears the brunt of CVD burden and has one of the highest risks of dying
from non-communicable diseases (NCDs) worldwide. There is currently
an epide
...
miological transition on the continent, where NCDs is projected
to outpace communicable diseases within the current decade. Unchecked
increases in CVD risk factors have contributed to the growing burden of three
major CVDs—hypertension, cardiomyopathies, and atherosclerotic diseasesleading to devastating rates of stroke and heart failure. The highest age
standardized disability-adjusted life years (DALYs) due to hypertensive heart
disease (HHD) were recorded in Africa. The contributory causes of heart failure
are changing—whilst HHD and cardiomyopathies still dominate, ischemic
heart disease is rapidly becoming a significant contributor, whilst rheumatic
heart disease (RHD) has shown a gradual decline. In a continent where health
systems are traditionally geared toward addressing communicable diseases,
several gaps exist to adequately meet the growing demand imposed by CVDs.
Among these, high-quality research to inform interventions, underfunded
health systems with high out-of-pocket costs, limited accessibility and
affordability of essential medicines, CVD preventive services, and skill
shortages. Overall, the African continent progress toward a third reduction
in premature mortality come 2030 is lagging behind. More can be done in
the arena of effective policy implementation for risk factor reduction and
CVD prevention, increasing health financing and focusing on strengthening
primary health care services for prevention and treatment of CVDs, whilst
ensuring availability and affordability of quality medicines. Further, investing
in systematic country data collection and research outputs will improve the accuracy of the burden of disease data and inform policy adoption on
interventions. This review summarizes the current CVD burden, important
gaps in cardiovascular medicine in Africa, and further highlights priority
areas where efforts could be intensified in the next decade with potential
to improve the current rate of progress toward achieving a 33% reduction
in CVD mortality.
more
Where There Are No Pharmacists
Sarah Andersson; Beverley Snell
Third World Network; Health Action International Asia Pacific
(2019)
C2
2nd edition. A Guide to Managing Medicines for All Health Workers. Available for purchase via following link https://www.twn.my/title2/books/HAI.htm
Where there are no trained pharmacists serving communities, other categories of health workers ar
...
e called upon to order, buy, store, dispense and advise people on rational use of medicines. Where There Are No Pharmacists explains how to order them, store them, prepare them, dispense them and use them safely and effectively - it is about managing medicines. Information to help communities benefit from the use of medicines is also included.
This book walks readers through each step, covering topics ranging from policy issues to patient education. It provides guidance for anyone who is doing the work of a pharmacist; anyone who sells, dispenses, prepares, manages, or explains to others how to use medicines.
more
These guidelines have been developed to provide guidance to the Ministry of Health in managing applications for registration of human pharmaceutical products in Rwanda. It was compiled by the Technical Working Group (TWG) on Medicines Evaluation and
...
Registration (MER) of the East African Community Medicine Regulatory Harmonization (EAC MRH) Project. The group relied on their experiences and knowledge on medicines registration requirements of their individual Countries. World Health Organization (WHO) and the International Conference on Harmonization of Technical Requirements of Medicines for Human Use (ICH) and other available literature.
more
The first important change is a new priority ranking of the available medicines for MDR-TB treatment, based on a careful balance between expected benefits and harms. Treatment success for MDR-TB is currently low in many countries. This could be incr
...
eased by improving access to the highest-ranked medicines for all patients with MDR-TB.
more
This guideline covers making people aware of how to correctly use antimicrobial medicines (including antibiotics) and the dangers associated with their overuse and misuse. It also includesmeasures to prevent and control infection that can stop peopl
...
e needing antimicrobials or spreadinginfection to others. It aims to change people's behaviour to reduce antimicrobial resistance and thespread of resistant microbes.
more
As a public good, antimicrobial medicines require rational use if their effectiveness is to be preserved. However, up to 50% of antibiotic use is inappropriate, adding considerable costs to patient care, and increasing morbidity and mortality. In ad
...
dition, there is compelling evidence that antimicrobial resistance is driven by the volume of antimicrobial agents used. High rates of antimicrobial resistance to common treatments are currently reported all over the world, both in health care settings and in the community. For over two decades, the Region of the Americas has been a pioneer in confronting antimicrobial resistance from a public health perspective. However, those efforts need to be stepped up if we are to have an impact on antimicrobial resistance and want to quantify said impact.
more
Global actions to reduce antimicrobial resistance (AMR) include optimising the use of antimicrobial medicines in human and animal health. In countries with weak healthcare regulation, this requires a greater understanding of the drivers of antibioti
...
c use from the perspective of providers and consumers. In Bangladesh, there is limited research on household decision-making and healthcare seeking in relation to antibiotic use and consumption for humans and livestock. Knowledge is similarly lacking on factors influencing the supply and demand for antibiotics among qualified and unqualified healthcare providers. The aim of this study is to conduct integrated research on household decision-making for healthcare and antibiotic use, as well as the awareness, behaviours and priorities of healthcare providers and sellers of antibiotics to translate into policy development and implementation
more
On 15–16 December 2020, WHO and the Medicines for Malaria Venture co-convened a technical consultation to consider the preferred product characteristics (PPCs) for drugs used in malaria chemoprevention. The main goal of the technical consultation
...
was to agree on the most important PPCs for drugs to protect populations from malaria (chemoprevention), while considering relevant measures of efficacy and the safety data needed to support WHO policy recommendations.
more
Licensed pharmaceutical products (marketing authorization) should be manufactured only by licensed manufacturers (holders of a manufacturing authorization) whose activities are regularly inspected by competent national authorities. This guide to GMP shall be used as a standard to justify GMP status,
...
which constitutes one of the elements of the WHO Certification Scheme on the quality of pharmaceutical products moving in international commerce, through the assessment of applications for manufacturing authorizations and as a basis for the inspection of manufacturing facilities. It may also be used as training material for government medicines inspectors, as well as for production, QC and QA personnel in the industry
more
Pharmaceutical regulators are at the forefront of ensuring that only safe and effective medicines are authorized and available in the market. This document builds on the recommendations in the above publication and has been prepared to specifically
...
assist national medicines regulatory authorities to understand the nature and extent of oxytocin quality issues and to provide key technical information and quality requirements for oxytocin products in dossier assessments. Furthermore, this document also presents recommendations on other regulatory actions needed to ensure that only quality-assured oxytocin products are authorized and made available to women.
more
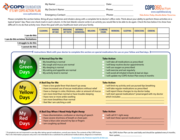
Please complete the section below. Bring all your medicines and inhalers along with a complete list to doctor’s office visits. Think about your ability to perform these activities on a typical “green” day. Place one check mark in each column.
...
In the last (blank) column write in an activity you would like to be able to do again. Check the box below it to show how difficult it is to do that activity now. Share this goal with you healthcare team and your family.
more
Using an inhaler is the most common and effective way of taking asthma medicines. If you’re NOT using your inhaler correctly, you might not be getting the full dose of medicine your doctor or asthma nurse prescribed because the medicine can’t re
...
ach your
lungs. Instead it might be hitting the back of your throat, orstaying on your tongue or in your mouth where it won’t help atall.
more
It is estimated that around 2 billion people worldwide do not have access to essential medicines. Access to medicines in the Eastern Mediterranean Region varies among countries, depending on their i
...
ncome level and allocation of domestic resources to medicine and vaccine procurement. Access to safe and effective antibiotics remains a major challenge, especially for low- and middleincome countries. Barriers to access include high prices for new products, weak regulatory systems, substandard and falsified antibiotics, shortages of essential antimicrobials and inefficient procurement and supply management systems.
more
Early-warning indicators to prevent stock-outs and overstocking of antiretroviral, antituberculosis and antimalaria medicines.
The fight against fake drugs by NAFDAC in Nigeria
The fight against fake drugs by NAFDAC in Nigeria Olike Chinwendu
KIT (Royal Tropical Institute) Development Policy & Practice Vrije Universiteit Amsterdam
(2008)
CC
Review over the work and challenges of the Nigerian National Agency for Food and Drug Administration and Control (NAFDAC) in combatting counterfeiting of medicines in Nigeria.