Filter
509
Text search:
nucleic
Acid
Featured
95
126
Language
Document type
204
153
65
46
18
7
6
4
2
1
1
1
Countries / Regions
31
18
17
12
11
7
7
6
6
6
6
5
5
4
4
4
4
3
3
3
3
3
3
3
3
3
2
2
2
2
2
2
2
2
2
2
2
2
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
Authors & Publishers
Publication Years
Category
117
94
33
14
13
12
3
Toolboxes
104
86
54
43
21
17
12
12
12
11
11
8
5
4
4
3
2
2
1
1
1
1
Available in Arabic, Chinese, English, French, Portuguese and Spanish
https://apps.who.int/iris/handle/10665/334254
Se estima que unos 10 millones de personas contrajeron tuberculosis (TB) en el 2019 y que a unos 3 millones de ellas no se les diagnosticó ni se les notificó la enfermedad. Con el propósito de poner fin a la epidemia mundial de la tuberculosis para el 2030, sigue siendo necesario ampliar la capac
...
Decontaminating the Ebola Facilities in Sierra Leone
Updated Januar 2016
Zika and dengue viruses remain significant public health threats. These viruses share the same Aedes (Stegomyia) mosquito vectors and geographic distributions but infections cannot be readily distinguished clinically and need to be differentiated from each other, and from other circulating arboviral
...
ECDC proposed case definition for surveillance of Zika virus infection
Information note.
This information note provides a strategic overview of key implementation considerations for diagnostic integration using these devices, and is primarily intended for use by national laboratory services and TB, HIV, and hepatitis programme managers.
It may also be of inte ...
This information note provides a strategic overview of key implementation considerations for diagnostic integration using these devices, and is primarily intended for use by national laboratory services and TB, HIV, and hepatitis programme managers.
It may also be of inte ...
Journal of Infectious Diseases and Therapeutics, 2013, 1, 17-24
Presently, there is no evidence that the virus responsible for the current COVID-19 pandemic is carried by domestic food-producing animals, such as chickens, ducks, other poultry, pigs, cattle, camels, horses, sheep, goats, rabbits, guinea pigs or fish. While live animals can be a source of pathogen
...
Laboratory biosafety guidance related to coronavirus disease (COVID-19): Interim guidance, 28 January 2021
recommended
The latest update (28 January 2021) includes the following addition and revision:
biosafety aspects for working with antigen-detecting rapid diagnostic test;
handling new variants of SARS-CoV-2 in the laboratory;
updated assay decontamination before disposal;
personal protectiv
...
This document aims to provide interim guidance for microbiology and virology experts, other laboratory professionals, laboratory managers, infectious disease programme managers, public health professionals and other stakeholders that provide primary, confirmatory or advanced testing for SARS-CoV-2,
...
Testing Guidance and Interpretation of Results for Healthcare Providers
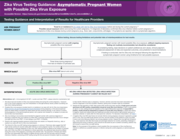
Zika Virus Testing Guidance: Asymptomatic Pregnant Women
with Possible Zika Virus Exposure
Testing Guidance and Interpretation of Results for Healthcare providers
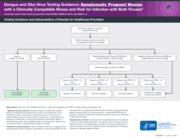
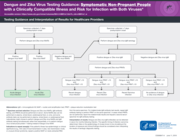
Testing Guidance and Interpretation of Results for Healthcare Providers Dengue and Zika Virus
This guidance synthesizes current evidence on dengue laboratory testing and diagnostics and provides practical recommendations for laboratories, clinicians, public health officials, and programme managers involved in dengue diagnosis, surveillance, and control, in the context of the global emergency
...
Tuberculosis (TB) remains a serious public health threat in South Africa and is the
leading cause of death in people living with HIV (PWH). Pre-hospital and early in-
hospital mortality is of particular concern in PWH. Significant progress has been made
to date, but a monumental effort is require
...