Filter
3723
Text search:
vaccine
Featured
431
866
Language
3318
250
184
105
105
87
48
44
16
15
11
10
10
7
6
6
6
5
5
5
5
5
5
5
5
5
5
5
5
4
4
4
4
4
4
3
3
3
3
3
2
2
2
2
2
2
2
2
2
1
1
1
1
1
1
1
1
1
1
1
1
1
1
Document type
1455
694
580
261
206
197
80
72
72
47
31
13
13
1
Countries / Regions
191
144
87
83
79
76
73
73
71
67
62
60
54
49
48
45
44
44
42
40
40
40
38
38
37
36
35
31
31
31
30
27
26
25
24
23
22
22
22
19
18
18
16
16
14
14
13
13
12
12
12
12
11
11
11
11
11
10
9
9
8
8
7
7
7
7
7
6
6
6
6
6
5
5
5
5
4
4
4
4
4
4
4
4
3
3
3
3
3
3
3
3
2
2
2
2
2
2
2
2
2
2
2
2
2
2
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
Authors & Publishers
698
152
119
102
85
85
59
53
51
42
40
39
38
37
34
30
29
24
24
17
17
17
16
15
14
14
14
14
13
13
13
13
12
11
11
11
11
10
10
10
10
10
10
10
10
10
9
9
9
9
9
8
8
8
8
8
8
8
8
8
8
8
8
8
8
8
7
7
7
7
7
7
7
7
7
7
7
7
7
7
7
7
7
7
6
6
6
6
6
6
6
6
6
6
6
6
6
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
5
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
4
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
3
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
2
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
Publication Years
1827
1655
223
17
1
Category
1446
316
181
148
131
37
34
1
Toolboxes
865
166
161
129
120
96
90
89
88
86
79
78
73
69
64
60
59
55
51
47
33
24
12
9
2
1
nterim guidance, first issued 25 January 2021, updated 15 June 2021, updated 19 November 2021, updated 23 February 2022, updated 18 August 2022
These WHO interim recommendations on the use of the Astra Zeneca – Oxford University AZD1222 vaccine against Covid-19 were developed on the basis of advice issued by the Strategic Advisory Group of Experts on Immunization (SAGE) and the evidence s
...
ummary included in the background document referenced below.
This document has been updated: Version 15 March 2022.
more
On the 9 February 2021, Africa CDC convened a special session of the Africa Task Force for COVID-19 to review existing data and evidence and recommend
8 February 2021
This document highlights 10 well established Risk Communication and Community Engagement (RCCE) principles that have proven their power. Together they put communities at the heart of the roll out of new vaccines, treatments and tests and promote trust, the critical ingredient for al
...
l community action. Informed, engaged and empowered communities are the bedrock for the arrival of new vaccines, treatments and tests that will be introduced to reduce the spread of COVID-19 and save lives. With communities fully engaged and actively participating through the full cycle of planning, delivery and assessment for biomedical tools, demand for these tools can be increased, leading to widespread and effective uptake and use
more
AstraZeneca-SKBio in South Korea and Serum Institute of India
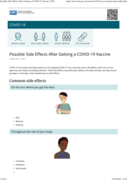
COVID-19 vaccination will help protect you from getting COVID-19. You may have some side effects, which are normal signs that your body is building protection. These side effects may affect your ability to do daily activities, but they should go away in a few days. Some people have no side effects.
...
Available in English, Spanish, Vietnamese, Chinese
more
Interim recommendations for use of the inactivated COVID-19 vaccine, CoronaVac, developed by Sinovac
These WHO interim recommendations for use of the Sinovac-CoronaVac were developed on the basis of advice issued by the Strategic Advisory Group of Experts on Immunization (SAGE) and the evidence summary included in the background document and annexes referenced below.
This document has been updat
...
ed: version 15 March 2022.
more
21 May 2021. he Practical Approach to Care Kit – PACK – is used by nurses, doctors and community health workers in their everyday work to care for patients at the clinic and in the community – PACK Adult, PACK Adolescent, PACK Child and PACK Community.
PACK Adult is also known as APC (Adult P
...
rimary Care) outside of the Western Cape. Both form part of the the Ideal Clinic initiative: www.idealhealthfacility.org.za
PACK Home is designed for people seeking more information about how to care for their own health and the health of their family at home.
Available in English, Afrikaans and isiXhosa
more
Since the emergence of COVID 19 in December 2019, various public health responses measures have been implemented to control the pandemic. Among measures taken by the Africa CDC was the launch of PACT initiative to accelerate COVID 19 testing. Key to the initiative is the engagement of Community Heal
...
th Workers (CHWs) in risk communication and community engagement (RCCE), surveillance activities for early case identification, contacts tracing and in facilitating referrals for testing and continuum of care.
As of 31 May 2021, Through PACT support, over 17154 CHWs have been trained and locally deployed in 24 AU Member states. The PACT supported CHWs visited more than 2,568,654 households for community engagement activities, active case search and contact tracing, identified 1,618,601 Contacts, 710,167 COVID 19 suspect cases based on the standard case definition and facilitated referrals for 553053 (78%) suspect cases for testing. These efforts were crucial for early identification and isolation of cases in limiting further transmission.
more
FEY ENFOMASYON POU MOUN K’AP RESEVWA AVEK MOUN K’AP BAY SWEN OTORIZASYON POU ITILIZASYON IJANS (EUA) POU VAKSIN MODERNA COVID-19 POU ANPECHE CORONAVIRUSDISEASE 2019
(COVID-19) NAN PERSONN 18 ANE AK PI GRAN
Vaksen DTaP (Difteri, Tetanòs, Koklich): Sa Ou Bezwen Konnen
Vaksen Td
Sa Ou Bezwen Konnen
This quick guide offers basic information about COVID-19, the Sinovac-CoronaVac COVID-19 vaccine and what to
expect following vaccination: ...
These WHO interim recommendations for use of the BBV152 COVAXIN vaccine were developed on the basis of advice issued by the Strategic Advisory Group of Experts on Immunization (SAGE) and the evidence summary included in the background document and a
...
nnexes referenced below.
This document has been updated: version 15 March 2022.
The vaccine is formulated from an inactivated SARS-CoV-2 antigen and is presented in single dose vials and multidose vials of 5, 10 and 20 doses.
more