Filter
2638
Filtered Results: 2638
Text search:
confirmación
Featured
Recommendations
291
New Publications
685
Language
Document type
No document type
1367
Guidelines
555
Studies & Reports
265
Strategic & Response Plan
156
Manuals
141
Fact sheets
52
Training Material
43
Situation Updates
30
Online Courses
10
Infographics
9
Brochures
5
Resource Platforms
4
Dashboards/Maps
1
Countries / Regions
India
124
Global
76
Congo, Democratic Republic of
76
Paraguay
65
Latin America and the Carribbean
62
South Africa
54
Colombia
52
Kenya
48
Senegal
46
Argentina
44
Western and Central Europe
42
Ethiopia
42
Nigeria
39
Sierra Leone
38
Rwanda
38
Africa
38
Liberia
38
Venezuela
33
Uganda
31
Malawi
27
Nepal
26
Brazil
26
Peru
25
Guinea
25
Burkina Faso
24
Bolivia
24
Tanzania
22
Zambia
22
Ghana
21
Bangladesh
21
Myanmar / Burma
21
Philippines
20
Eastern Europe
20
Ecuador
20
Madagascar
20
Benin
19
Cameroon
18
Chile
17
Haiti
17
El Salvador
16
Syria
16
East and Southern Africa
16
Mali
16
Zimbabwe
15
West and Central Africa
15
Ukraine
14
Central African Republic
13
South–East Asia Region
12
Spain
12
Côte d’Ivoire / Ivory Coast
12
Namibia
12
Middle East and North Africa
10
Togo
9
Mozambique
9
Niger
8
South Sudan
8
Lesotho
7
Western Pacific Region
7
Botswana
7
Guatemala
7
Cambodia
6
Asia
6
Germany
6
Russia
6
Honduras
6
Somalia
6
Pakistan
6
Mexico
5
Yemen
5
Angola
5
Chad
5
Gambia
4
Guinea-Bissau
4
Eastern Europe and Central Asia
4
Indonesia
4
Eswatini/ Swaziland
4
China
3
Afghanistan
3
Jordan
3
North America
3
Uruguay
3
France
3
Nicaragua
3
North Macedonia
2
Timor Leste/ East Timor
2
Turkey
2
Iran
2
Albania
2
Burundi
2
Morocco
2
Dominican Republic
2
United Kingdom
2
Lebanon
2
Sri Lanka
2
Sudan
2
Palestine
2
Georgia
2
Tajikistan
1
Thailand
1
Kazakhstan
1
Laos
1
Estonia
1
Uzbekistan
1
Malaysia
1
Vanuatu
1
Mauritania
1
USA
1
Cape Verde
1
Hungary
1
Mauritius
1
Tunisia
1
Portugal
1
Southern Africa
1
Oman
1
Vietnam
1
Moldova
1
Poland
1
Iraq
1
Cuba
1
Authors & Publishers
Publication Years
Category
Countries
1168
Clinical Guidelines
297
Key Resources
90
Public Health
82
Pharmacy & Technologies
71
Women & Child Health
65
Capacity Building
42
Toolboxes
COVID-19
337
TB
287
Rapid Response
217
NTDs
167
HIV
138
Ebola & Marburg
136
Mental Health
72
Malaria
71
Zika
57
Cholera
54
AMR
42
Caregiver
40
Conflict
32
Polio
32
Pharmacy
31
Refugee
24
Natural Hazards
23
NCDs
23
Disability
21
Planetary Health
14
Specific Hazards
10
Global Health Education
8
2.0 Rapid Response
5
Health Financing Toolbox
3
Typhoon
2
Social Ethics
1
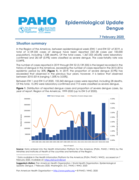
Epidemiological Update
Dengue
7 February 2020
Situation summary
In the Region of the Americas, between epidemiological week (EW) 1 and EW 521 of 2019, a total of 3,139,335 cases of dengue have been reported (321.58 cases per 100,000 population), including 1,538 deaths. Of the total cases, 1,367,...
A Training Course for Service Providers
The report will describe how the Ebola Response MPTF, which has attracted contributions from 38 Member States, one business and many individuals, has offered a transparent and strategic tool to support the Ebola response. As of 31 January 2015, the Fund had total pledges amounting to US$142 million,...
Early detection, assessment and response to acute public health events:
The purpose of this publication is to to provide a practical, stepwise approach to the implementation of the national action plans on AMR within the human health sector; and to provide a process and collation of existing WHO tools to prioritize, cost, implement, monitor and evaluate national action ...
The standards of care cover the routine care and management of complications occurring for women and their babies during labour, childbirth and the early postnatal period, including those of small babies during the first week of life. They define priorities for improving the quality of maternal and ...
Expert opinion of the European Tuberculosis Laboratory Initiative core group members for the WHO European Region.
These guidelines have been developed to provide guidance to the Ministry of Health in managing applications for registration of human pharmaceutical products in Rwanda. It was compiled by the Technical Working Group (TWG) on Medicines Evaluation and Registration (MER) of the East African Community M...
Specific measures are being taken within the National Tuberculosis Control Programme (NTP) to address the MDR TB problem through appropriate management of patients and strategies to prevent the propagation and dissemination of MDR TB.
The term "Programmatic Management of Drug Resistant TB" (PMD...
The term "Programmatic Management of Drug Resistant TB" (PMD...
hese are two parallel guidelines, one for small hospitals and another one for large hospitals. In view of heavy burden of malaria and prevalence of drug resistant falciparum malaria in the South-East Asia Region, the guidelines were developed for use by medical personnel who treat severe malaria pat...