Filter
488
Text search:
medical
circumcision
Featured
47
157
Language
Document type
313
63
55
17
16
10
7
2
1
1
1
Countries
35
31
30
28
24
19
17
14
13
13
13
13
12
8
8
6
5
5
4
4
4
3
2
2
2
2
2
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
1
Authors & Publishers
Publication Years
Category
290
31
28
20
7
3
2
Toolboxes
125
15
9
8
6
4
3
2
2
2
2
1
1
1
1
1
1
1
The "Safe Male Circumcision Community Mobilization Training: A Guide for Facilitators in Uganda" is
a training manual designed to equip community mobilizers with the skills to promote safe male circumcisi
...
The objective of this project was to list the medical devices required to provide the essential reproductive, maternal, newborn and child health interventions defined by existing WHO guidelines and publications, in order to improve access to these d
...
Technical brief. This brief provides a description of urethral fistula, addresses the risk factors and potential practices that could lead to a fistula, diagnosis, first management and response, and prevention measures to reduce these complications through improved understanding by practitioners. It
...
The WHO Framework for the clinical evaluation of devices for male circumcision
outlines a multi-phase process to ensure the safety, efficacy, and acceptability of new devices, especially for use in resource-limited settings
The Protection of Personal Information (POPI) Act
Confidentiality: providing and protecting information
• Health care practitioners hold information about patients that is private and sensitive.
• The National Health Act (Act No. 61 of 2003) provides that this information must not be given to
...
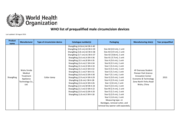
The List of WHO-prequalified Male Circumcision Devices contains male circumcision devices for adult male populations, appropriate for use in resource-limited settings
Transforming Health: Accelerating attainment of Health Goals | THE SECOND MEDIUM TERM PLAN FOR HEALTH
Large File: 12 MB!
Updated to Reflect the 2009 Medical Eligibility Criteria of the World Health Organization
The procurement of essential medicines and other health products is a critical function in support of the effective discharge of WHO’s mandate, and WHO values the importance of the quality of essential medicines and health products that are supplied to countries. The first World Health Assembly in
...
Vol. 2: Clinicians' Guide
Clinicians’ Guide is designed to assist busy medical practitioners in the field with patient management by providing current, essential, practical guidance and background, packaged into a single resource
Clinicians’ Guide is designed to assist busy medical practitioners in the field with patient management by providing current, essential, practical guidance and background, packaged into a single resource
These guidelines are designed for ICRC and other health professionals – nurses, midwifes, doctors – who either lack experience in antenatal care or are not used to working in countries where medical infrastructure is underdeveloped or non-existe
...
This handbook is for health care providers involved in the care of girls and women who have been subjected to any form of female genital mutilation (FGM). This includes obstetricians and gynaecologists, surgeons, general medical practitioners, midwi
...
In: Antenatal Care, Maternal Care, Under 5 Clinics, Family Planning Clinics and HIV Exposed Child Follow Up.
This 5th Edition of the Malawi Guidelines for Clinical Management of HIV in Children and Adults is implemented from January 2022. It replaces all previous editions of the Malawi Antiretrovir
...